At a glance
Cancers are composed not only of tumour cells, but also the tumour microenvironment (TME), which consists of many different types of normal cells and the extracellular matrix (ECM) - a network of proteins and carbohydrates that provides structure to and controls the development and function of tissues. The TME co-evolves with cancer cells and strongly influences all steps of progression, from the earliest stages to the development of metastasis and therapeutic resistance. Among the most important components of the TME are various cells of the immune system. While they are initially a barrier to cancer formation, cancer cells orchestrate a complex network of interactions within the TME to escape surveillance and elimination by immune cells, and even co-opt some immune cells to drive their growth and progression. The composition and physical properties (such as stiffness) of the ECM also change dramatically during tumour progression, providing important cues that control the behaviour of cancer and normal cells within the TME.
The GCI has been instrumental in revealing the importance of the TME during tumour development, establishing it as a source of powerful biomarkers and therapeutic targets with great potential. Recent findings have also shown that the TME mediates the influence of broader physiological and environmental factors, such as diet and bodyweight, on cancer initiation, progression, and therapeutic response. Using advanced technology, GCI investigators study how the TME evolves and reveal the mechanisms underlying its influence on cancer biology and outcomes. We have developed innovative genetically engineered models to study the TME and unique methods to recreate it in the laboratory, allowing us to understand how it functions at a fundamental level. Combining these approaches with our strong connections to the clinic allows us to discover new precision medicine strategies harnessing the tumour microenvironment to improve cancer therapy.
Areas of Focus
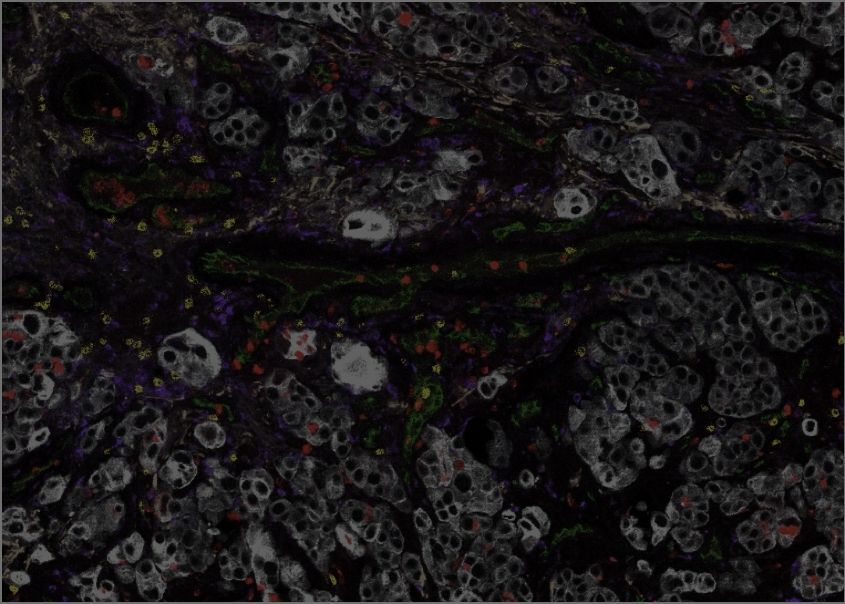
Tumour Immunology and Immunotherapy. By harnessing the power of the patient’s immune system to attack and destroy tumours, immunotherapies can induce highly durable responses, and even cures, in some cancer types that typically have a very poor prognosis. Nonetheless, these revolutionary new therapies are affected by the same issues that have limited other precision medicine strategies to date — subsets of tumours are non-responsive, and resistance develops in most cases where an initial response is achieved. Research at the GCI focuses on discovering the mechanisms that cancers use to resist immunotherapy and identifying biomarkers that can empower clinical decision-making, allowing the patients who will experience the best responses to receive immunotherapy while those whose tumours are primed to resist can be spared from unnecessary side effects. We use our powerful technology for spatially mapping “-omics” data to reveal the topography of the immune response within cancer tissue, identifying the key features differentiating immunotherapy response from resistance and uncovering powerful predictive biomarkers. Our research programs seek to identify the next generation of immunotherapy targets among the networks of tumour/immune cell interactions, and to discover strategies to enhance immunotherapy by leveraging the influence of diet, the microbiome, and the duration and timing of treatment, including comparing the effects of continuous and intermittent dosing and defining the role of chronobiology (the mechanisms underlying time-related phenomena such as circadian rhythms) in immunotherapy response.
Innate Immunity, Inflammation and Cancer. A complex biological response known as inflammation is a normal part of the body’s response to injury, its defences against infection and the wound healing process, among other important roles. It involves specific populations of immune cells, particularly those referred to as innate immune cells, as well as changes in the vasculature and the ECM, and a range of growth factors, cytokines and other mediators of intercellular communication. Inflammation typically ends as the source of injury is removed. However, tumour development triggers a sustained inflammatory response known as chronic inflammation, characterized by different populations of innate immune cells, a different spectrum of soluble factors, and different effects on tissue structure and stromal cells compared to acute inflammatory responses. Tumours can actively promote chronic inflammation by releasing molecules such as cytokines, in addition to damaging local tissue as they grow. Chronic inflammation promotes tumour growth by stimulating the development of a blood supply (angiogenesis), interfering with immune responses that suppress early cancers, and providing signals that stimulate tumour cell proliferation and survival. Health conditions that promote systemic and chronic inflammation predispose toward cancer development and can accelerate tumour progression. GCI investigators use genetic models and advanced analysis of patient samples to understand the complex relationships between the innate immune system, inflammation and cancer. Working within national and international networks including those funded by Cancer Research UK, the Brain Tumour Funders’ Collaborative, and the EU-funded TRANSCAN program, we are mapping the innate immune and inflammatory responses to a range of poor-outcome cancers to reveal new therapeutic strategies targeting cancer-associated inflammation.
Team members
Our Discoveries
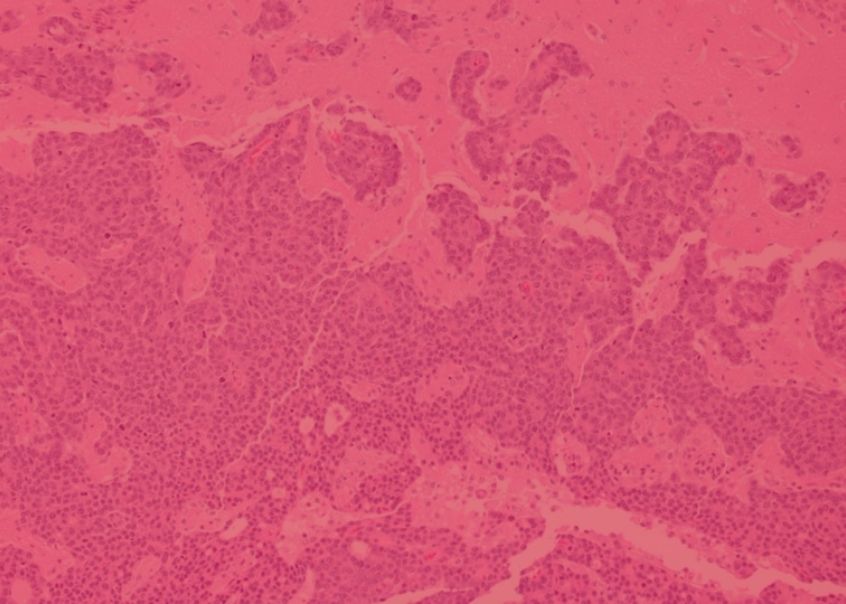
GCI research was vital in establishing the importance of the microenvironment in breast cancer. We were among the first to establish that the microenvironment is a source of biomarkers that can predict breast cancer outcome, and our cutting-edge imaging and analysis has revealed the spatial organization of the immune response in breast cancer, including the presence of subtypes of the immune microenvironment that are associated with specific features of breast cancer biology and can predict clinical outcomes.
Finak, G. et al. Stromal gene expression predicts clinical outcome in breast cancer. Nat Med. 2008 May;14(5):518-27. doi: 10.1038/nm1764. Epub 2008 Apr 27. PMID: 18438415
Saleh SMI, et al. Identification of Interacting Stromal Axes in Triple-Negative Breast Cancer. Cancer Res. 2017 Sep 1;77(17):4673-4683. doi: 10.1158/0008-5472.CAN-16-3427. Epub 2017 Jun 26. PMID: 28652250
Gruosso, T. et al. Spatially distinct tumor immune microenvironments stratify triple-negative breast cancers. J Clin Invest. 2019 Apr 1;129(4):1785-1800. doi: 10.1172/JCI96313. Epub 2019 Mar 18. PMID: 30753167
Through our leading roles in clinical and translational research networks, we are using the GCI’s advanced platforms for spatially resolved, single-cell analysis to reveal the organization of the immune response in human cancer in unprecedented detail. Studies led by GCI investigators have used this technology to find spatial relationships and cellular “neighborhoods” that correlate with responses and outcomes in metastatic melanomas and recurrent lung cancers treated with immunotherapy. These discoveries are leading to innovative new biomarkers that will dramatically improve precision medicine, allowing clinicians to maximize the benefits of immunotherapy for those patients who are primed to respond, while sparing others from unnecessary side effects.
Moldoveanu D, et al. Spatially mapping the immune landscape of melanoma using imaging mass cytometry. Sci Immunol. 2022 Apr;7(70):eabi5072. doi: 10.1126/sciimmunol.abi5072. Epub 2022 Apr 1.PMID: 35363543
Sorin, M. et al., Single-cell spatial landscape of immunotherapy response reveals mechanisms of CXCL13 enhanced antitumor immunity. J Immunother Cancer. 2023 Feb;11(2):e005545. doi: 10.1136/jitc-2022-005545. PMID: 3672508
GCI investigators are among the world’s leaders in understanding the cancer-associated roles of myeloid cells (cells of the innate immune system that both form and mature within the bone marrow). An international project under our leadership, supported by the Brain Tumour Funders’ Collaborative, used the GCI’s unique single-cell spatial analysis platform to reveal that the presence of a unique population of macrophages, a type of myeloid cell, was associated with better clinical outcomes in brain cancer. Work done at the GCI has also shown that myeloid cells known as neutrophils are crucial mediators of breast cancer metastasis to the lungs in the context of obesity. We have discovered metabolic properties of neutrophil subpopulations that support breast cancer metastasis to the liver, and with our Faculty members and partners we are revealing how neutrophil extracellular traps (NETs), DNA- and protein-containing structures released by neutrophils, are involved in tumour progression. This work is leading to promising new therapeutic strategies to disrupt the cancer-promoting functions of myeloid cells.
Karimi E, et al. Single-cell spatial immune landscapes of primary and metastatic brain tumours. Nature. 2023 Feb;614(7948):555-563. doi: 10.1038/s41586-022-05680-3. Epub 2023 Feb 1. PMID: 36725935
McDowell SAC, et al. Neutrophil oxidative stress mediates obesity-associated vascular dysfunction and metastatic transmigration. Nat Cancer. 2021 May;2(5):545-562. doi: 10.1038/s43018-021-00194-9. Epub 2021 May 3. PMID: 35122017
Hsu BE, et al. Immature Low-Density Neutrophils Exhibit Metabolic Flexibility that Facilitates Breast Cancer Liver Metastasis. Cell Rep. 2019 Jun 25;27(13):3902-3915.e6. doi: 10.1016/j.celrep.2019.05.091.PMID: 31242422
Rayes RF, et al. Primary tumors induce neutrophil extracellular traps with targetable metastasis promoting effects. JCI Insight. 2019 Jul 25;5(16):e128008. doi: 10.1172/jci.insight.128008. PMID: 31343990
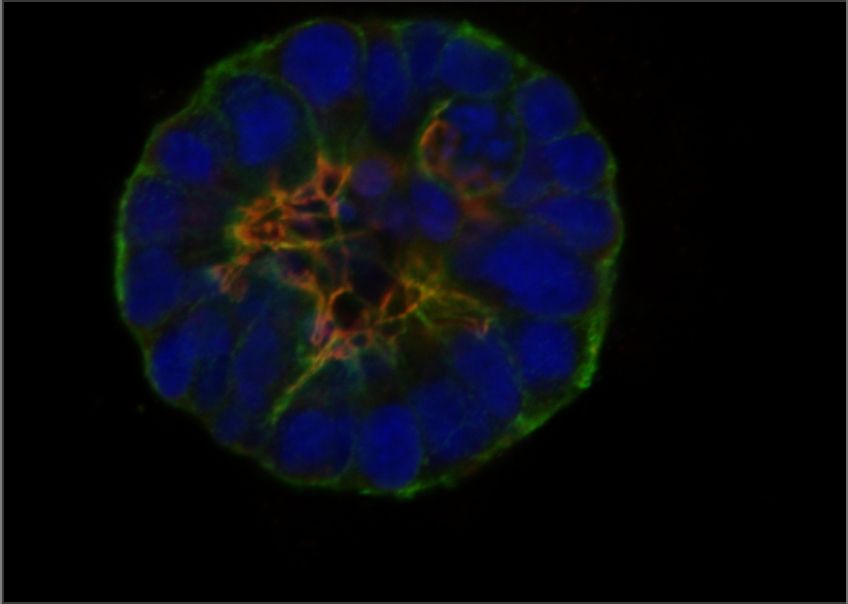
One of the most important ways that cells interact with their microenvironment is through a class of proteins called integrins, which recognize and bind to specific components of the extracellular matrix (ECM). Integrins have critical functions in cancer cells, where they transduce signals from the environment that influence cell growth and division, survival, and important steps of metastasis. GCI investigators were among the first to use genetic engineering to reveal the involvement of integrin proteins in breast cancer progression, including their role in determining whether emerging cancers progress or enter a state of growth arrest known as dormancy. We have also developed sophisticated new approaches, in collaboration with chemical and biological engineers, to model cancer-associated changes in the ECM in the laboratory, allowing us to study the fundamental mechanisms linking these changes with cancer progression.
White, D. et al. Targeted disruption of beta1-integrin in a transgenic mouse model of human breast cancer reveals an essential role in mammary tumor induction. Cancer Cell. 2004 Aug;6(2):159-70. doi: 10.1016/j.ccr.2004.06.025. PMID: 15324699
Bui, T. et al. Functional Redundancy between β1 and β3 Integrin in Activating the IR/Akt/mTORC1 Signaling Axis to Promote ErbB2-Driven Breast Cancer. Cell Rep. 2019 Oct 15;29(3):589-602.e6. doi: 10.1016/j.celrep.2019.09.004. PMID: 31618629
Bui, T. et al. Emergence of β1 integrin-deficient breast tumours from dormancy involves both inactivation of p53 and generation of a permissive tumour microenvironment. Bui T, Gu Y, Ancot F, Sanguin-Gendreau V, Zuo D, Muller WJ. Oncogene. 2022 Jan;41(4):527-537. doi: 10.1038/s41388-021-02107-7. Epub 2021 Nov 15. PMID: 34782719
Mok S, et al. Mapping cellular-scale internal mechanics in 3D tissues with thermally responsive hydrogel probes. Nat Commun. 2020 Sep 21;11(1):4757. doi: 10.1038/s41467-020-18469-7. PMID: 32958771