Immune checkpoint inhibitors have revolutionized cancer treatment by harnessing the power of the body’s immune system. However, many patients do not respond to available therapies, highlighting an urgent need to identify new immunosuppressive pathways and therapeutic targets. We discovered that production of adenosine by the CD39-CD73 axis is an important immune checkpoint that promote cancer. With support from the Canadian Institutes of Health Research, we are defining the molecular ecosystem governing adenosine-mediated effects in cancer. Specifically, we study the impact of the adenosine production via CD39 and CD73 and transport across ENT1 and ENT2 in various immune cell populations.
We also leverage large multi-omics datasets to identify new mechanisms of immunotherapy resistance. We work closely with collaborators of the Terry Fox Institute Marathon-of-Hope Cancer Centers network, the Francis Crick Institute and Roche’s immunotherapy Centers of Research Excellence (imCORE). Our consortium allows us to investigate, at an unprecedented level, the landscape of genomic, transcriptomic and proteomic associations with immunotherapy clinical responses.
What we are
currently
working on
With partners in Belgium, France and Germany, and with our GCI collaborator Dr Morag Park, we are mapping the adaptation of triple-negative breast cancer to chemo-immunotherapy. This project leverages spatial transcriptomics and multiplex analysis of primary and metastatic lesions from 3 large randomized clinical trials.
We recently discovered that blocking or deleting ENT1, the major regulator of extracellular adenosine concentrations, significantly enhanced CD8+ T cell-dependent anti-tumor immunity. Using gene-targeted mice and preclinical cancer models, we are now characterizing the cell-specific function of ENT1 and ENT2 in various immune cell populations.
Using bioinformatics, we discovered Protease-activated receptor-2 (PAR2) as a potential new target to overcome resistance to immune checkpoint inhibitors. PAR2 is a receptor activated by specific tumor proteases and involved in pathophysiological inflammation. We are investigating the impact of PAR2 on tumor immunity using gene-targeted mice and proteomics analysis of human tumors, and will identify patient populations that may benefit from PAR2-targeted therapy.
With collaborators at CHUM and Ottawa Hospital Research Institute, we are defining the immune landscape and functional interactions at single-cell level of rare ovarian cancers. Our goal is to identify new biomarkers for diagnosis and new treatment trajectories, including immunotherapies and antibody-drug conjugates.
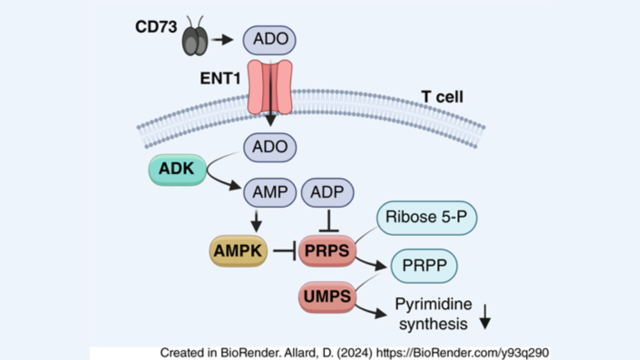
- ENT1 : une nouvelle cible thérapeutique Nous avons étudié l’effet du transporteur équilibratif de nucléosides-1 (ENT1), principal régulateur des concentrations extracellulaires d’adénosine, sur l’immunité antitumorale. Le blocage ou la délétion d’ENT1 chez l’hôte a significativement renforcé les réponses antitumorales dépendantes des lymphocytes T CD8+. Les tumeurs greffées chez des souris déficientes en ENT1 présentaient une infiltration accrue de lymphocytes T CD8+ effecteurs, dotés d’un profil transcriptomique cytotoxique renforcé. Sur le plan mécanistique, la captation d’adénosine médiée par ENT1 inhibait l’activité de la phosphoribosyl-pyrophosphate synthétase dans les lymphocytes T activés, supprimant ainsi la production d’uridine-5′-monophosphate et de ses dérivés nécessaires à la synthèse de l’ADN et de l’ARN. ENT1 as a new Target We investigated the effect on tumor immunity of equilibrative nucleoside transporter-1 (ENT1), the major regulator of extracellular adenosine concentrations. Blocking or deleting host ENT1 significantly enhanced CD8+ T-cell–dependent antitumor responses. Tumors inoculated into ENT1-deficient mice showed increased infiltration of effector CD8+ T cells with an enhanced cytotoxic transcriptomic profile. Mechanistically, ENT1-mediated adenosine uptake inhibited the activity of phosphoribosyl pyrophosphate synthetase in activated T cells, thereby suppressing production of adenosine receptors suppress immune cells. uridine 5′-monophosphate and its derivatives required for DNA and RNA synthesis.
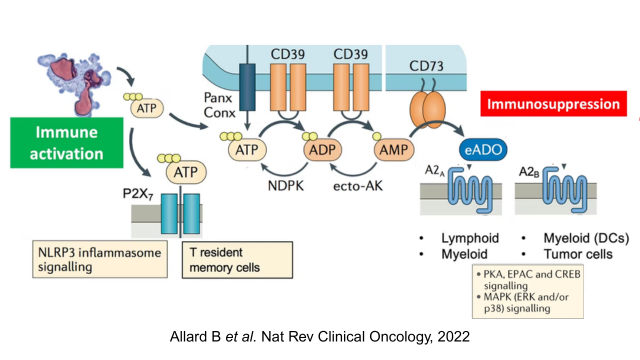
- Voie de l'adénosine On sait depuis près d'un demi-siècle que l'adénosine (ADO) module la fonction immunitaire. Les niveaux d'ADO extracellulaire sont contrôlés par l'hydrolyse de l'adénosine triphosphate extracellulaire par des ecto-nucléotidases telles que CD39 et CD73. La signalisation de l'adénosine nuit à la fonction de diverses cellules immunitaires, y compris les cellules T, les cellules NK et les cellules myéloïdes. L'activation de la PKA dépendante de l'AMPc et le blocage des voies NF-KB et JAK-STAT sont des mécanismes généralisés par lesquels les récepteurs A2A et A2B de l'adénosine suppriment les cellules immunitaires. The Adenosine pathway Adenosine (ADO) has been known to modulate immune function for nearly half a century. Levels of extracellular ADO are controlled by the hydrolysis of extracellular adenosine triphosphate by ecto-nucleotidases such as CD39 and CD73. Adenosine signaling impairs the function of a variety of immune cells, including T cells, NK cells and myeloid cells. Activation of cAMP-dependent PKA and blockade of NF-KB and JAK-STAT pathways are generalized mechanisms by which A2A and A2B adenosine receptors suppress immune cells.
- Professor, Rosalind and Morris Goodman Cancer Institute, McGill University, Faculty of Medicine and Health Sciences, Department of Microbiology and Immunology
- Associate Professor, faculty of pharmacy, Université de Montréal
Our team
Major discoveries
- 2025 Adenosine Uptake through the Nucleoside Transporter ENT1 Suppresses Antitumor Immunity and T-cell Pyrimidine Synthesis
- 2025 The Terry Fox Research Institute Marathon of Hope Cancer Centres Network: A pan-Canadian precision oncology initiative
- 2025 Targeting G Protein-Coupled Receptors in Immuno-Oncological Therapies
- 2023 CD73 Inhibits cGAS–STING and Cooperates with CD39 to Promote Pancreatic Cancer
- 2023 Adenosine A2A receptor is a tumor suppressor of NASH-associated hepatocellular carcinoma
- 2022 Leveraging big data of immune checkpoint blockade response identifies novel potential targets






.png)
.png)
.png)